Is Stem Cell Legal in Malaysia?
In order to answer the above question, we have extracted below the relevant information from The STAR and the Malaysian Guidelines for Stem Cell Research and Therapy 2009, so that you can understand and make a better informed decision.

Guidelines for Cell and Gene Therapy Products
According to The Star, Guidelines for cell and gene therapy products (CGTPs) issued by the Health Ministry will be enforced from 2021, says Dr Lee Boon Chye (Deputy Health Minister).
MACT (Malaysian Association for Cell Therapy) president Dr Lim Teck Onn said the effective use of stem cell for medical purposes was evidence-backed, but cell therapy might be controversial for aesthetic applications.
“The use of stem cell for genuine medical therapy has never really been an issue. There are data and proven trials and even US Food and Drug Administration-approved indications. The big problem however is its use in wellness, anti-aging and some beauty products. As an industry association, we have an obligation to make sure that companies involved in these activities do so responsibly and not take patients and consumers for a ride,” he said.
To download the whole Malaysian Guidelines for Cell and Gene Therapy Products 2016, please click here.

Guidelines for Cell and Gene Therapy Products
According to The Star, Guidelines for cell and gene therapy products (CGTPs) issued by the Health Ministry will be enforced from 2021, says Dr Lee Boon Chye (Deputy Health Minister).
MACT (Malaysian Association for Cell Therapy) president Dr Lim Teck Onn said the effective use of stem cell for medical purposes was evidence-backed, but cell therapy might be controversial for aesthetic applications.
“The use of stem cell for genuine medical therapy has never really been an issue. There are data and proven trials and even US Food and Drug Administration-approved indications. The big problem however is its use in wellness, anti-aging and some beauty products. As an industry association, we have an obligation to make sure that companies involved in these activities do so responsibly and not take patients and consumers for a ride,” he said.
To download the whole Malaysian Guidelines for Cell and Gene Therapy Products 2016, please click here.
Guidelines for Stem Cell Therapy in Malaysia
Haemopoietic stem cell and umbilical cord stem cell transplantations are the most established form of stem cell therapy. The use of other stem cells including hES (human embryonic stem cells) and somatic stem cells is considered experimental. Xenotransplantation or therapies involving the use of animal stem cells or animal cells are currently prohibited.
Indications for stem cell/cell-based therapies
1. Standard/established
(a) The current indications for HSC therapy (HSCT) are listed in the National Guidelines for Haemopoietic Stem Cell Therapy.
2. Potential/development/experimental
The following applications shall either be at the in vitro, animal studies or clinical trial settings;-
(a) The use of HSC, HSC-derived cells umbilical cord stem cells in tissue repair, regeneration and vascularisation shall be considered developmental.
(b) MSC (mesenchymal stem cell) has shown potential in chronic inflammatory diseases, graft rejection and graft versus host disease (GVHD) and shall be done in research settings.
(c) The use of other types of stem cells e.g. neural stem cells in Parkinson’s disease, cardiac stem cells, hepatic stem cells, pancreatic stem cells, skeletalmuscle stem cell, stem cells of skin, lungs, retinal and intestinal epithelium as well as inducible pluripotent stem cells are still experimental.
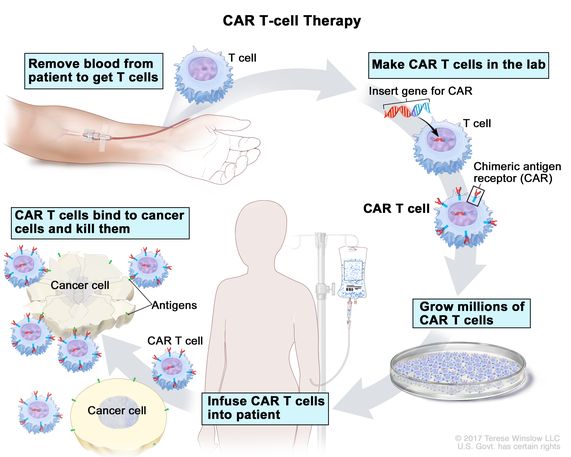
(d) Cell-based therapies e.g. antigen-specific T-cells and dendritic cells shall be done in research settings.
(e) Gene therapy to correct genetic disorder e.g. subacute combined immune deficiency disorders (SCID) and thalassaemia is still in developmental phase. The use of lentiviral shall be carried out in a P3 laboratory.
(f) All other indications not listed in as standard or established shall be considered experimental and must first be approved by the institutional Guidelines For Stem Cells Therapy review board(IRB) and/or institutional ethics committee. Copies of the proposal must also be submitted to the NSCRE sub-committee.
3. Therapy using human embryonic stem cells
(a) The use of hESC for therapy shall be considered experimental.
4. Xenotransplantation
(a) Xenotransplantation involving stem cells/cells e.g. islet cells for tissue repair and regeneration shall not be performed until more scientific and clinical evidence (Phase 3) is obtained.
(b) Rejection and the risks of transmission of infectious diseases have not been adequately dealt with. (c) Clinical trials shall occur only when there are preclinical data indicating a high probability of benefit to the recipients and data on safety.
(d) Any clinical xenotransplantation shall only be approved by the NSCRE subcommittee.
5. Patient evaluation
Please refer to the National Guidelines for Haemopoietic Stem Cell Therapy.
6. Regulation and Standards
(a) Private healthcare facilities and services intending to perform or performing stem cell or cell based therapies shall be licensed under the Private Healthcare Facility and Services Act 1998.
(b) It is recommended that these centres perform internal and external audits to ensure quality, viability, purity, safety, reproducibility and efficacy of the end-products.
(c) The procurement and processing of stem cells shall comply with the National Standards of Procurement and Processing of stem cells.
(d) Laboratories performing gene therapy research involving the use of viral vectors shall comply with the Biosafety Level 3.
(e) Personnel performing stem cell transplants shall be adequately trained and proficient and shall acquire privileging status from the respective institutions.
To download the whole Malaysian Guidelines for Stem Cell Research and Therapy 2009 guidelines, please click here.
Related:

Comments
Post a Comment