9 Best Stem Cell Therapy Clinics in US and the World 2025
This article explores leading stem cell centers within the U.S. and
worldwide.
The global regenerative medicine market size is calculated at USD 20.09
billion in 2024 and is expected to be worth USD 139.70 billion by 2034,
expanding at a CAGR of 21.4% from 2024 to 2034, as a result of the rising
demand for cell and gene therapies. (source)
The global stem cell therapy market size (sub-set of regenerative medicine)
was exhibited at USD 14.15 billion in 2023 and is projected to hit around
USD 49.11 billion by 2033, growing at a CAGR of 13.25% during the forecast
period 2024 to 2033. (Nova1Advisor)
Below, we cover leading stem cell centers. Each one has treated large
populations of patients with adult stem cells. At least one (Regenexx) is
maintaining a Patient Registry to document long-term patient outcomes.
Others, like GIOSTAR, have been working with the U.S. FDA on clinical trials
related to the treatment of COVID-19.
Related: Best Stem Cell Transplant Hospitals
Each of GIOSTAR’s clinics is licensed for the application of stem cell therapy. Since 2000, its team of scientists and clinicians have been developing and utilizing stem cell-based clinical protocols for the purpose of stem cell treatment.
Within the United States, GIOSTAR treats patients at its Chicago, IL, location. It also serves clients at its clinical sites based in Mexico, India, Brazil, Thailand and U.A.E.
GIOSTAR is one of the world’s most established stem cell treatment providers, having treated thousands of patients over the past 21 years. The company has also been working with the U.S. FDA on trials for the treatment of COVID-19.
The stem cells clinic uses stem cell therapies to treat various ailments, including the following:
Clinical Trials Results
After the completion of this clinical study, there was an improvement in MS patient disability. The 1-month mark of the study documented improvements in mobility, hand, bladder, bowel, and sexual functions. Importantly, the study demonstrated that a sustained one-year umbilical cord stem cell therapy has more durable benefits than current MS drug therapies.
HI-STEM Research Groups:
Nonetheless, to meet the immediate needs of its clients, the company also has taken steps to meet the requirements of the FDA and COFEPRIS, a Mexican institution equivalent to FDA in MSCs import and export.
Celltex also works with Mexican hospitals that are established and certified that allowed the company’s cell-banking clients to receive their stem cells for medical purposes.
QC Kinetix offers various non-surgical pain management therapies to help patients who are dealing with all types of conditions, such as: knee pain, joint pain, shoulder pain, elbow pain, sciatic nerve pain, and much more.
There is no one-size fits all solution for stem cell therapy. Whether it's allogeneic or autologous stem cells, we will need to assess every individual and pre-qualify them for a personalised recommendation.
If you need more information about stem cell treatment, feel free to contact us.
Treatment
What possible benefits can the patient get from stem cell therapy? How can these be measured and how long will it take?
Are there other medications or special care the patient needs aside from the treatment?
Which stem cell source best fits the patient’s condition?
What are other treatment options for the patient’s condition?
What is their stem cell procedure (from harvesting to follow-up check-up)?

Several of them, have been treating patients for over a decade.
Please note that we are not advising patients to seek treatment from these centres. We are identifying and featured them to allow readers to seek out more information.1. GIOSTAR
The Global Institute of Stem Cell Therapy and Research (GIOSTAR)
provides adult stem cells for autologous and allogeneic stem cell
therapy, based on research by
Dr. Anand Srivastava. This stem cell treatment group offers treatments for a range of
chronic conditions, musculoskeletal injuries, and degenerative
diseases.
GIOSTAR can treat joint pain (knees, shoulders, hips), orthopedic
injuries, arthritis, lung diseases, hair loss, erectile dysfunction,
autoimmune diseases, and post-COVID complications. It can also provide
anti-aging and aesthetic treatments, including IV vitamin therapy.
Each of GIOSTAR’s clinics is licensed for the application of stem cell therapy. Since 2000, its team of scientists and clinicians have been developing and utilizing stem cell-based clinical protocols for the purpose of stem cell treatment.
Within the United States, GIOSTAR treats patients at its Chicago, IL, location. It also serves clients at its clinical sites based in Mexico, India, Brazil, Thailand and U.A.E.
GIOSTAR is one of the world’s most established stem cell treatment providers, having treated thousands of patients over the past 21 years. The company has also been working with the U.S. FDA on trials for the treatment of COVID-19.
2. Stem Cell Institute (Panama)
Founded by Dr. Neil Riordan, a globally recognized stem cell expert and visionary, the Stem Cell Institute in Panama is among the world’s leaders in stem cell research and therapy. Their treatments focus on well-targeted combinations of allogeneic umbilical cord stem cells, as well as autologous bone marrow stem cells.The stem cells clinic uses stem cell therapies to treat various ailments, including the following:
- spinal cord injury (SCI)
- rheumatoid arthritis
- heart failure
- osteoarthritis
- multiple sclerosis
- autoimmune diseases
- genetic disorders, such as autism and cerebral palsy
Stem Cell Therapy for MS
One of their most recent studies exhibited the clinical feasibility of stem cell transplant process as a safe and effective treatment approach for patients with multiple sclerosis (MS).
Published in the Journal of Translational Medicine, the study
showed that umbilical cord stem cells can slow down MS disease
progression and decrease the frequency of flare-ups.
However, these stem cells did not exhibit the ability to repair damaged nerve cells or myelin sheaths.
Related: Why Stem Cell Transplants for Multiple Sclerosis Work so Well
However, these stem cells did not exhibit the ability to repair damaged nerve cells or myelin sheaths.
Related: Why Stem Cell Transplants for Multiple Sclerosis Work so Well
Clinical Trials Results
After the completion of this clinical study, there was an improvement in MS patient disability. The 1-month mark of the study documented improvements in mobility, hand, bladder, bowel, and sexual functions. Importantly, the study demonstrated that a sustained one-year umbilical cord stem cell therapy has more durable benefits than current MS drug therapies.
See what Tony Robbins has to say about his stem cell treatments at Stem
Cell Institute clinic in Panama: CLICK HERE
and
HERE.
6. Stem Cell Therapy
— Craig Brockie (@craigbrockie) March 30, 2025
Finally, Tony explored the incredible benefits of stem cell therapy.
• Repairs damaged tissues
• Rejuvenates the body
• Slows aging
You gotta listen to this… pic.twitter.com/mSiiTYEt2Y
3. HI-STEM: The Heidelberg Institute for Stem Cell Technology and Experimental Medicine
HI-STEM gGmbH is a non-profit public-private partnership between the
German Cancer Research Center (DKFZ)
in Heidelberg, Germany and the
Dietmar Hopp Stiftung.
HI-STEM performs cutting-edge research on normal and malignant stem cells
with the aim of translating these results into novel clinical
applications. This includes the development of novel diagnostic tools and
innovative therapies to monitor and target leukemic and solid tumor stem
cells as well as metastatic disease. HI-STEM was founded in 2008 and is
located on the fourth floor of the main building of the DKFZ.
They are an international team of more than sixty scientists, including group leaders, postdoctoral fellows, PhD, MD and MSc. students as well as technical staff. This research team focuses on three major topics and is directed by the managing director of HI-STEM, Professor Dr. Andreas Trumpp, as well as seven additional group leaders.
They are an international team of more than sixty scientists, including group leaders, postdoctoral fellows, PhD, MD and MSc. students as well as technical staff. This research team focuses on three major topics and is directed by the managing director of HI-STEM, Professor Dr. Andreas Trumpp, as well as seven additional group leaders.
HI-STEM Research Groups:
- Hematopoietic and Leukemic Stem Cells (Andreas Trumpp)
- Junior Group: Stem Cells at the Interface of Hematopoiesis, Immunity and Cancer (Simon Haas)
- Junior Group: Pattern Recognition and Digital Medicine (Daniel Hübschmann)
- Cancer Progression and Metastasis (Rene Jackstadt)
- Experimental Hematology (Mick Milsom)
- Inflammatory Stress in Stem Cells (Marieke Essers)
- Chronically progressive neurodegenerative diseases (Marc Thier)
- Cancer Stem Cells and Metastasis (Martin Sprick and Andreas Trumpp)
If you need more information about stem cell treatment in Germany, feel
free to contact us.
Regenexx Stem Cell Technology
The Regenexx® technology involves a procedure in which a small bone marrow sample is extracted through a needle and blood is drawn from a vein in the arm. These samples are then processed in a laboratory and the cells they contain are injected into the area needing repair, with the goal of delivering large numbers of stem cells to the site of injury.
Beyond FDA-Approved Cell Therapy
Regenexx is also a licensed offshore clinic in the Cayman Islands where patients can undergo treatments that utilize laboratory-expanded (“ex vivo”) stem cell populations. This approach allows for a much larger number of stem cells to be administered to the patient than is supported by U.S. law, which currently prohibits laboratory procedures that the FDA considers to exceed “minimal manipulation.”
Dr. Christopher Centeno is the Founder and CEO of Regenexx. He is a global authority in the culture expansion and clinical use of adult stem cells to treat orthopedic injuries and the visionary behind the Regenexx® technology.Cade Hildreth is also a Regenexx patient. Click here to read her experience.
DVC Partner Lab, Vitro Biopharma
DVC Stem sources its cells from its partner lab Vitro Biopharma Inc, a state of the art, US-based, FDA registered, cGMP compliant and ISO 9001 certified laboratory, and only use American Association of Tissue Bank (AATB) certified suppliers of full-term, ethically U.S.-donated human umbilical cords.
Led by Medical Director Louis A. Cona, MD, DVC Stem’s focus is on cord-tissue derived allogeneic stem cell therapy for degenerative conditions, wear and tear, and intelligent aging.
Notable patients the company has treated include Lou Ferrigno (champion bodybuilder/actor), Michael Armand Hammer (businessman/philanthropist), and David Lyons (Founder, MS Fitness Challenge), and others .
Okyanos maintains both a North American Office in Clearwater, FL, and a purpose-built Cell Therapy Surgical Center in Freeport, Grand Bahama. Okyanos’ stem cell treatments are performed in their state-of-the-art surgical centers under the care of board-certified doctors.
Okyanos is also fully licensed and regulated under the Bahamas Stem Cell Therapy and Research Act and adheres to U.S. surgical center standards. Click here to read our interview with Matthew Feshbach, Co-Founder and CEO of Okyanos.
Patients can then use their stored stem cells for regenerative purposes
through infusions or injections performed by a licensed physician.
Because the FDA considers an individual’s stem cells a drug if they have been expanded in large quantities, Celltex has begun the process of undertaking clinical trials on stem cells as a treatment for a range of medical conditions, seeking approval from the FDA to allow physicians to utilize these cells.
4. Regenexx
Headquartered in Denver, Colorado, Regenexx offers self-derived (autologous transplant) same-day stem cell treatments to patients with orthopedic injuries and conditions. Regenexx clinics incorporate a variety of regenerative approaches, drawing patients from all over the U.S. who are seeking innovative, non-surgical treatments.Regenexx Stem Cell Technology
The Regenexx® technology involves a procedure in which a small bone marrow sample is extracted through a needle and blood is drawn from a vein in the arm. These samples are then processed in a laboratory and the cells they contain are injected into the area needing repair, with the goal of delivering large numbers of stem cells to the site of injury.
Beyond FDA-Approved Cell Therapy
Regenexx is also a licensed offshore clinic in the Cayman Islands where patients can undergo treatments that utilize laboratory-expanded (“ex vivo”) stem cell populations. This approach allows for a much larger number of stem cells to be administered to the patient than is supported by U.S. law, which currently prohibits laboratory procedures that the FDA considers to exceed “minimal manipulation.”
Dr. Christopher Centeno is the Founder and CEO of Regenexx. He is a global authority in the culture expansion and clinical use of adult stem cells to treat orthopedic injuries and the visionary behind the Regenexx® technology.Cade Hildreth is also a Regenexx patient. Click here to read her experience.
5. DVC Stem
Headquartered in Seven Mile Beach, Grand Cayman, physicians at DVC Stem have been using complimentary alternative therapies to clinically treat patients with various medical conditions. It is one of the most advanced stem cell clinics in the Caribbean, with IRB-approved protocols, a fully licensed staff, and a facility that is accredited by the medical governing bodies of the Cayman Islands.DVC Partner Lab, Vitro Biopharma
DVC Stem sources its cells from its partner lab Vitro Biopharma Inc, a state of the art, US-based, FDA registered, cGMP compliant and ISO 9001 certified laboratory, and only use American Association of Tissue Bank (AATB) certified suppliers of full-term, ethically U.S.-donated human umbilical cords.
Led by Medical Director Louis A. Cona, MD, DVC Stem’s focus is on cord-tissue derived allogeneic stem cell therapy for degenerative conditions, wear and tear, and intelligent aging.
Notable patients the company has treated include Lou Ferrigno (champion bodybuilder/actor), Michael Armand Hammer (businessman/philanthropist), and David Lyons (Founder, MS Fitness Challenge), and others .
6. Okyanos
Founded in 2011, Okyanos is a stem cell therapy provider that specializes in treating patients with congestive heart failure (CHF) and other chronic degenerative conditions. Okyanos Cell Therapy uses internationally-approved technology to deliver a mixed population of fat (adipose) derived stem and regenerative cells (ADRCs) to patients with conditions such as the following:- cardiovascular disease
- orthopedic issues
- neurological disorders
- urological indications
- autoimmune conditions
Okyanos maintains both a North American Office in Clearwater, FL, and a purpose-built Cell Therapy Surgical Center in Freeport, Grand Bahama. Okyanos’ stem cell treatments are performed in their state-of-the-art surgical centers under the care of board-certified doctors.
Okyanos is also fully licensed and regulated under the Bahamas Stem Cell Therapy and Research Act and adheres to U.S. surgical center standards. Click here to read our interview with Matthew Feshbach, Co-Founder and CEO of Okyanos.
7. Celltex
Celltex specializes in cryopreserving mesenchymal stem cells (MSCs) for therapeutic use. Celltex acquires stem cells by collecting a small fat sample from a patient, from which MSCs are extracted, isolated, multiplied, and stored for future use (known as cell banking). Patients can then use their stored stem cells for regenerative purposes through infusions or injections performed by a licensed physician.Because the FDA considers an individual’s stem cells a drug if they have been expanded in large quantities, Celltex has begun the process of undertaking clinical trials on stem cells as a treatment for a range of medical conditions, seeking approval from the FDA to allow physicians to utilize these cells.
Nonetheless, to meet the immediate needs of its clients, the company also has taken steps to meet the requirements of the FDA and COFEPRIS, a Mexican institution equivalent to FDA in MSCs import and export.
Celltex also works with Mexican hospitals that are established and certified that allowed the company’s cell-banking clients to receive their stem cells for medical purposes.
8. QC Kinetix
QC Kinetix
is the place to go when you've been told invasive surgery is your best
option, or after you've tried everything to eliminate the pain and
immobility of your condition without success. With
multiple locations
throughout the United States, QC Kinetix provides comprehensive
regenerative medicine injection treatments. While not all patients may
be candidates for all therapies, they will help guide you to make the
most appropriate decisions for your particular condition. They treat
everyone from weekend warriors and professional athletes to active aging
patients who want to stay active.
QC Kinetix offers various non-surgical pain management therapies to help patients who are dealing with all types of conditions, such as: knee pain, joint pain, shoulder pain, elbow pain, sciatic nerve pain, and much more.
9. Stem Cell Malaysia (SCM)
The Malaysian Stem Cell Therapy Working Group (SCM) is a network of
specially trained multi-disciplined medical doctors and specialists,
providing advanced, research-driven, regenerative medicine, integrative
medicine and lifestyle medicine in Asia.
There is no one-size fits all solution for stem cell therapy. Whether it's allogeneic or autologous stem cells, we will need to assess every individual and pre-qualify them for a personalised recommendation.
The Malaysian Stem Cell Therapy Working Group (SCM), was established
in 2017 as a one-stop Stem Cell Therapy service provider assisting
patients worldwide seeking for Stem Cell therapy through our panel
of specialised doctors with years of experience in this field having
treated hundreds of patients in the past decade.
SCM operates in Gleneagles Hospital Kuala Lumpur, their main partnered medical centre in Malaysia. Gleneagles Hospital Kuala Lumpur is currently the leading private hospital in Malaysia's metropolitan capital.
SCM operates in Gleneagles Hospital Kuala Lumpur, their main partnered medical centre in Malaysia. Gleneagles Hospital Kuala Lumpur is currently the leading private hospital in Malaysia's metropolitan capital.
WhatsApp us for a Free Tele-Consultation: +60 12 - 877 2669
If you need more information about stem cell treatment, feel free to contact us.
Questions to Ask Before Undergoing a Stem Cell Therapy
Although these leading stem cell centers have built a good reputation in regenerative medicine, it is still important to probe these clinics before the proper procedure. These questions should cover what to expect from the treatment, safety and emergencies, cost, and the patient’s rights.Treatment
What possible benefits can the patient get from stem cell therapy? How can these be measured and how long will it take?
Are there other medications or special care the patient needs aside from the treatment?
Which stem cell source best fits the patient’s condition?
What are other treatment options for the patient’s condition?
What is their stem cell procedure (from harvesting to follow-up check-up)?
Safety and Emergencies
If the patient is given donor stem cells, what are the success and
failure rates of the therapy?
Are there possible long-term side effects of the treatment?
In case a side effect develops, what do stem cell centers do, who will the patient reach out to, and how immediate can they respond to the side effect?
How experienced are the doctor and the support staff who will perform the procedure?
How advanced and sterilized is the clinic’s equipment?
Costs
Are there possible long-term side effects of the treatment?
In case a side effect develops, what do stem cell centers do, who will the patient reach out to, and how immediate can they respond to the side effect?
How experienced are the doctor and the support staff who will perform the procedure?
How advanced and sterilized is the clinic’s equipment?
Costs
- What are the expenses for stem cell therapy?
- What other costs will the patient incur?
- What are the patient’s rights (confidentiality, update for new information, and the right to withdraw from the procedure)?
- What compensation will the patient be entitled to in case they will get injured while on the treatment process?
This is a condensed and adapted version of the original article that was published here.
Related: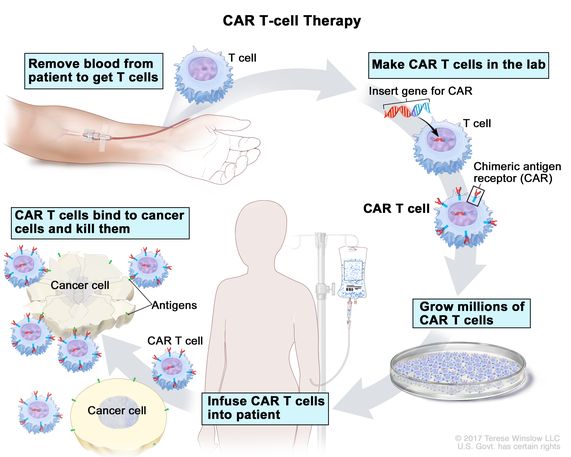

Comments
Post a Comment